BIO-PROSPERA CLINICAL RESEARCH PRIVATE LIMITED
Innovation For Better Tomorrow
- Proven Expertise
- Trusted & Connected

Driven by Discovery
BIO-PROSPERA CLINICAL RESEARCH PRIVATE LIMITED
We are end to end clinical trials service provider supporting comprehensive drug development and product lifecycle management. Our clients and partners include pharmaceutical, biotechnology, medical device, and government organizations absolute best in the industry.

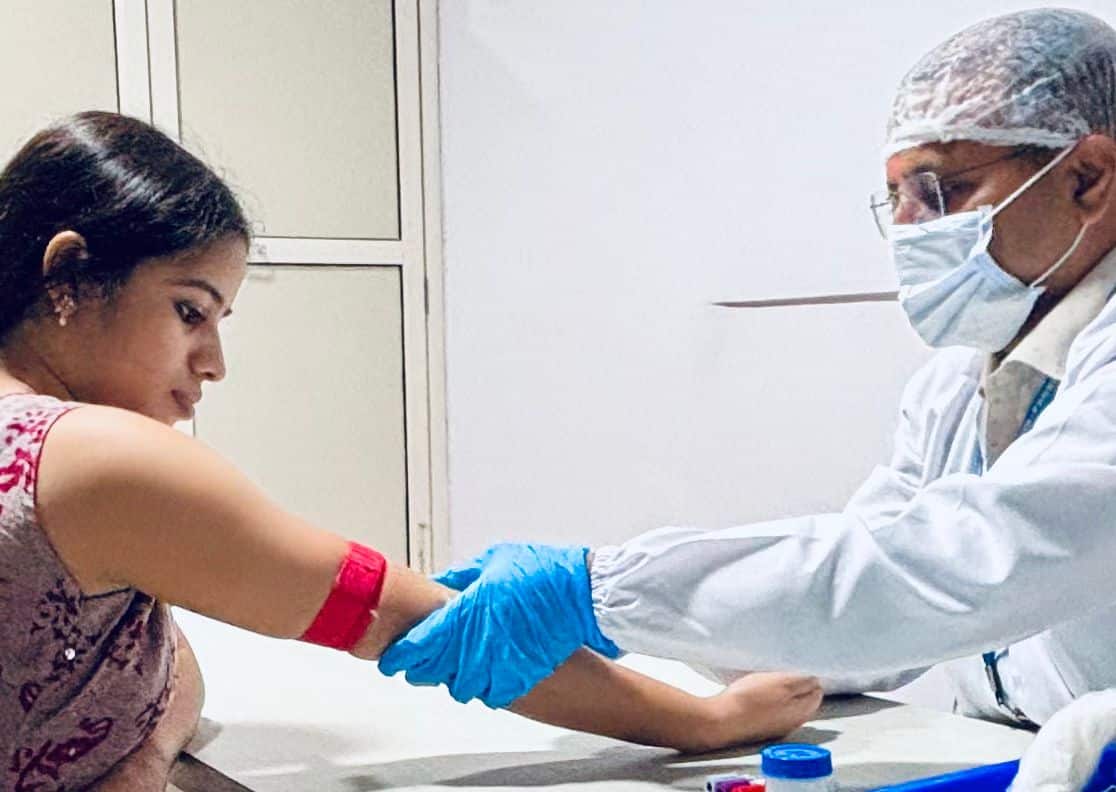



Driven by Data, Powered by Purpose
We deliver ethical, efficient clinical research backed by global reach and proven expertise.
Integrated Research Services
Full-spectrum trial support from design to regulatory submission across diverse therapeutic areas.
Therapeutic Areas We Serve
Specialized knowledge across therapeutic areas to support your clinical development.
Worldwide Support, Anytime
Reliable, round-the-clock support through our worldwide site network and vendor partnerships.
Why Choose Us
Redefining clinical research Support with Expertise and Reliability
It starts with our aim and as an adaptable Clinical Research Organisation (CRO) our expert networks of knowledgeable clinical research team can provide comprehensive assistance to support your clinical trials globally.
-
Proven Expertise
Global clinical trial support by experienced professionals from start to finish.
-
Trusted & Connected
ISO certified, award-winning, and globally connected across key markets.
our services
Comprehensive Clinical Research Services
We provide end-to-end clinical trial services India, delivering reliable solutions from study design to final submission. Our expertise includes clinical trial services for global market, ensuring compliance with international standards. With strong capabilities in clinical trial site management, we oversee recruitment, monitoring, and operational excellence.
Our team specializes in clinical trials protocol development tailored to regulatory needs, backed by robust processes for GCP-compliant clinical trial services. From early-stage studies to late-phase research, we also conduct Phase I clinical trials India, supporting pharmaceutical and biotech companies in bringing safe, effective therapies to market worldwide.
What Sets Us Apart
Bio-Prospera Strength
Our team has deep domain expertise in managing health care, pharmaceuticals, life sciences, technology services.
Bio-prospera is lead by a dedicated, experienced and ICH-GCP certified senior management team with significant knowledge of the pharmaceuticals, medical devices, nutraceuticals, Ayurveda and biotechnology sectors.
-
Dedicated team.
-
Excellent state of art facility.
-
ICH-GCP certified staff.
Testimonials
What Our Customers Say
“They guided us clearly through clinical trial stages. Their team is approachable, experienced, and committed to quality work.”

